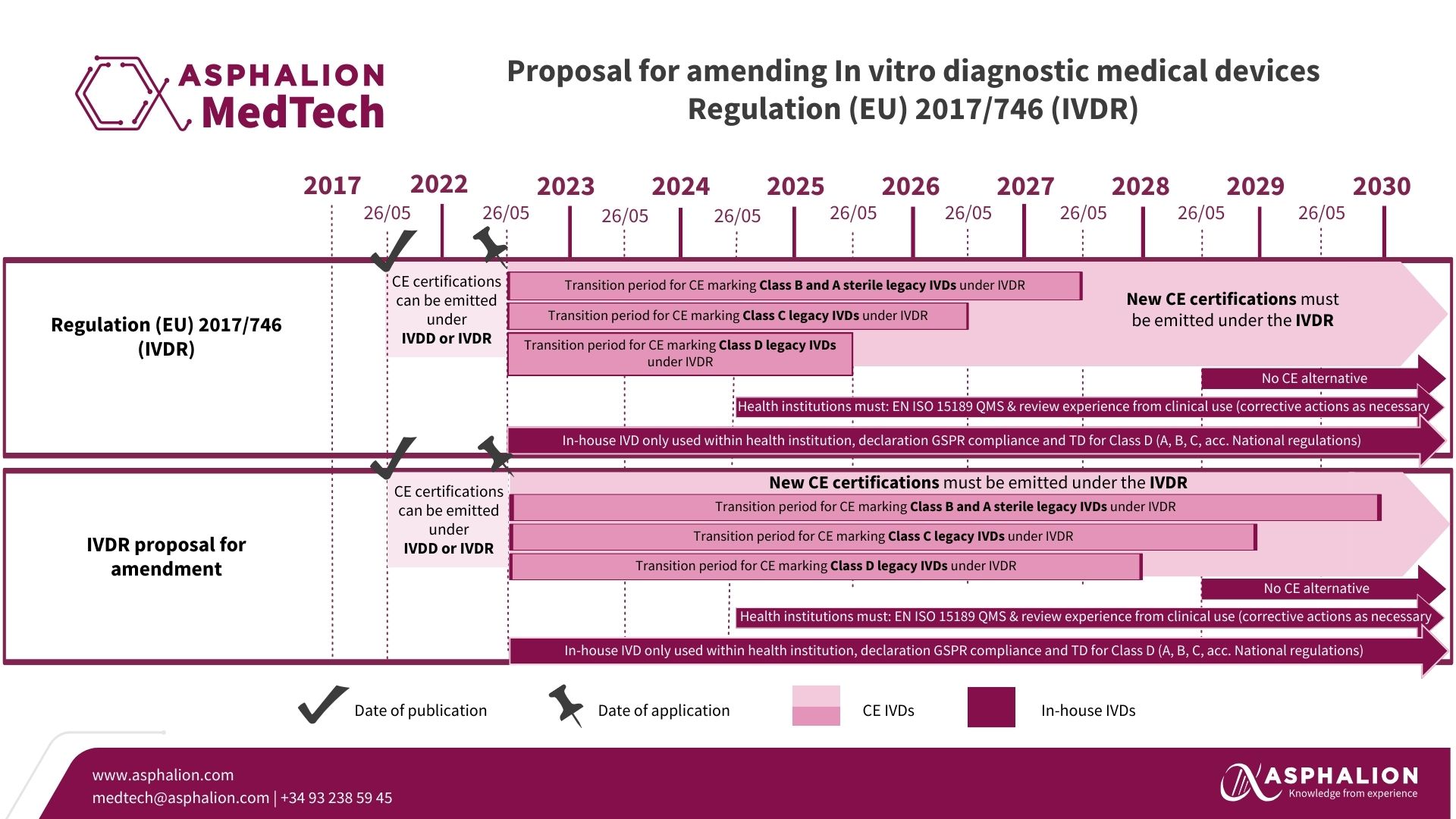
The European Commission took yesterday a significant step to ensure the continued availability of In Vitro Diagnostic medical devices (IVD) in the European market. In response to the considerable number of IVDs yet to comply with the In Vitro Diagnostic Medical Devices Regulation (EU) 2017/746 (IVDR), the Commission is proposing an extension of the transition periods.
This initiative echoes the previous adjustments made for Regulation (EU) 2017/745 (MDR) with Regulation (EU) 2023/607, highlighting the Commission’s commitment to prevent potential shortages of essential in vitro diagnostic tools.
The proposed amendment is a pivotal move to maintain the balance between regulatory compliance and the accessibility of vital diagnostic products for healthcare providers and patients across Europe.
Stay informed with the latest regulatory changes to navigate the evolving landscape of medical device regulations.
At Asphalion, our team of seasoned MedTech experts can help you in all your projects!
Contact us at: [email protected]
Source: AMENDING REGULATIONS EU 2017/745 & EU 2017/746