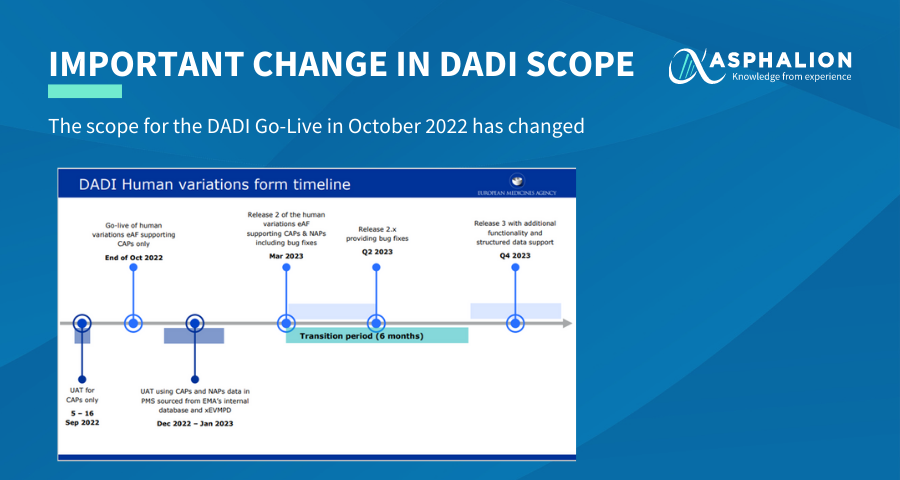
Due to the complexity in synchronisation of the data between xEVMPD and PMS, the scope of the Go-Live of October 2022 has been reduced to Centrally Authorized Products (CAPs) only.
The October release is considered a first major release and would be followed by subsequent releases in 2023.
One of those releases would include the support of the Nationally Authorized Products (NAPS, including National Procedures, Mutual Recognition Procedure and Decentralised Procedure) and it is expected for March 2023.
1st release: CAPs in October 2022
2nd release: NAPs in March 2023
Transition period (6 months) to start on March 2023
3rd release: Q4 2023 would support structured data and have additional functionalities
You can check here the complete new: eSubmission: Projects (europa.eu)
For further information, you can contact us at: [email protected]