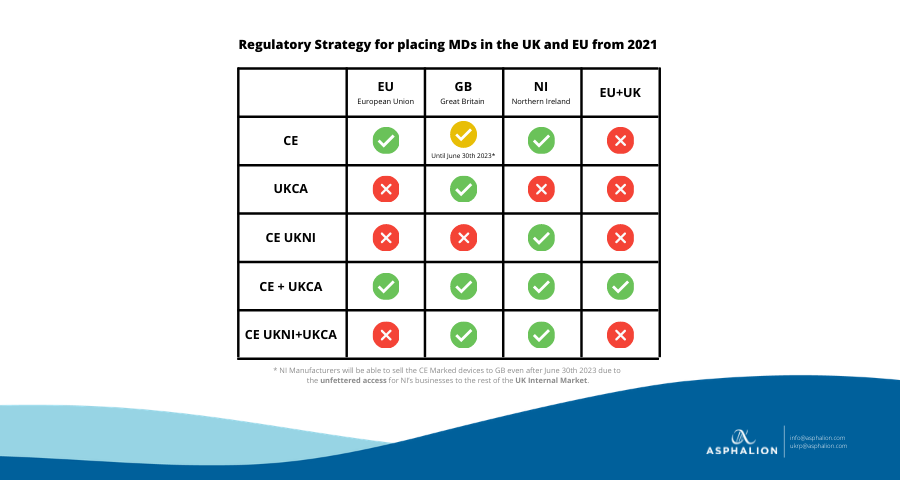
After the UK left the European Union on December 31st, 2020, the UK has defined two different legal frameworks for Great Britain and Northern Ireland.
- Great Britain will recognize the EU CE Marking until June 30th 2023; the new UKCA marking according to the UK MDR 2002 regulations, although already available, will be mandatory as of July 1st 2023 in GB.
- Northern Ireland will keep in line with the EU, meaning that the CE Marking will fully apply. However, a new marking has been introduced: CE UKNI according to the EU Legal Framework but only recognized in NI.
Nevertheless, two new requirements are common for both territories:
– Non-UK manufacturers shall have a UK Responsible Person based in the UK.
– All devices placed in the UK need be registered with the MHRA. For non-UK manufacturers, this registration is to be performed by the UKRP.
ASPHALION UK is a UK Responsible Person that can act as your UKRP and help you figure out which is the best strategy for your company to access the UK market.
For further information, you can contact us at: [email protected] and [email protected]