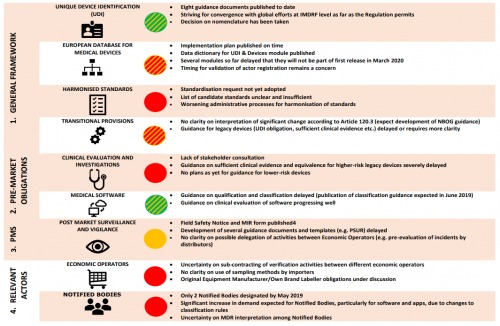
On 6th June 2019 COCIR (European Coordination Committee of the Radiological, Electromedical and healthcare IT Industry), the European trade association representing the medical imaging, radiotherapy, health ICT and electromedical industries, has published a document summarizing the current status in MDR implementation, which still shows little progress in a number of critical aspects. This is shown graphically on a diagram on page 3 of the document.
According to COCIR, “This is a critical period for the entire medical devices regulatory system. If the European Union’s ambition for medical devices are to be realised, the full regulatory framework will have to be in place and functioning. At this moment, it appears that much effort is needed to complete the framework in time. The current delays in implementation make the European Union a less attractive region for investment by medical device manufacturers, and likely increase cost for hospitals and threaten patient access.”
Asphalion closely follows news and movements around the new MDR and IVDR to better assess manufacturers and stakeholders on their implementation.
If you want to have more information, please contact [email protected]