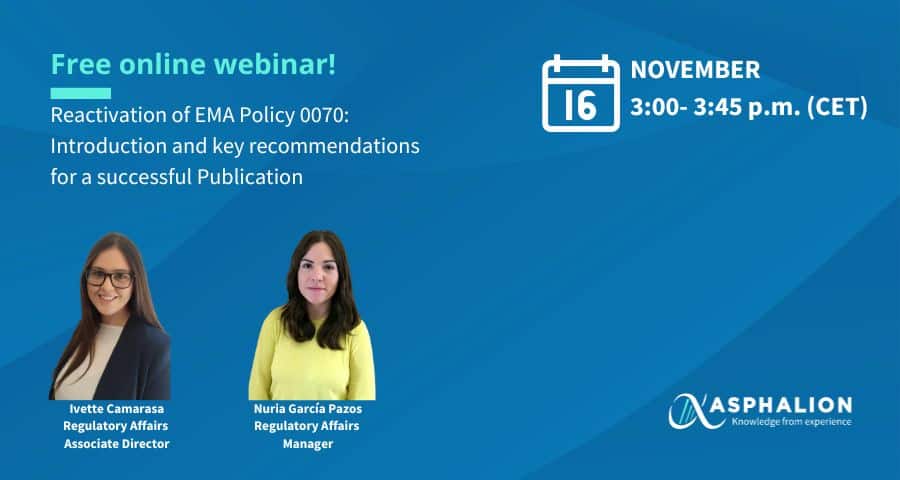
EMA Transparency Policy 0070, on the publication of Clinical Data, has been reinstated by EMA this year for new active substances registered under the centralized procedure, becoming effective from this September.
The publication of Clinical Data must follow strategies and techniques defined by the EMA, in order to assure the balance between the protection of personal data whilst maintaining the maximum data utility.
Join Asphalion experts Ivette Camarasa and Nuria García Pazos in this webinar, who will review the latest updates on the policy, timelines, CCI challenges and pre-submission meeting recommendations. They will go over:
- Latest updates on the policy,
- Publication and review timelines,
- CCI challenges
- Pre-submission meeting recommendations
Registrations are open. You can register here: TRANSPARENCY WEBINAR
For further information you can contact us at: [email protected]