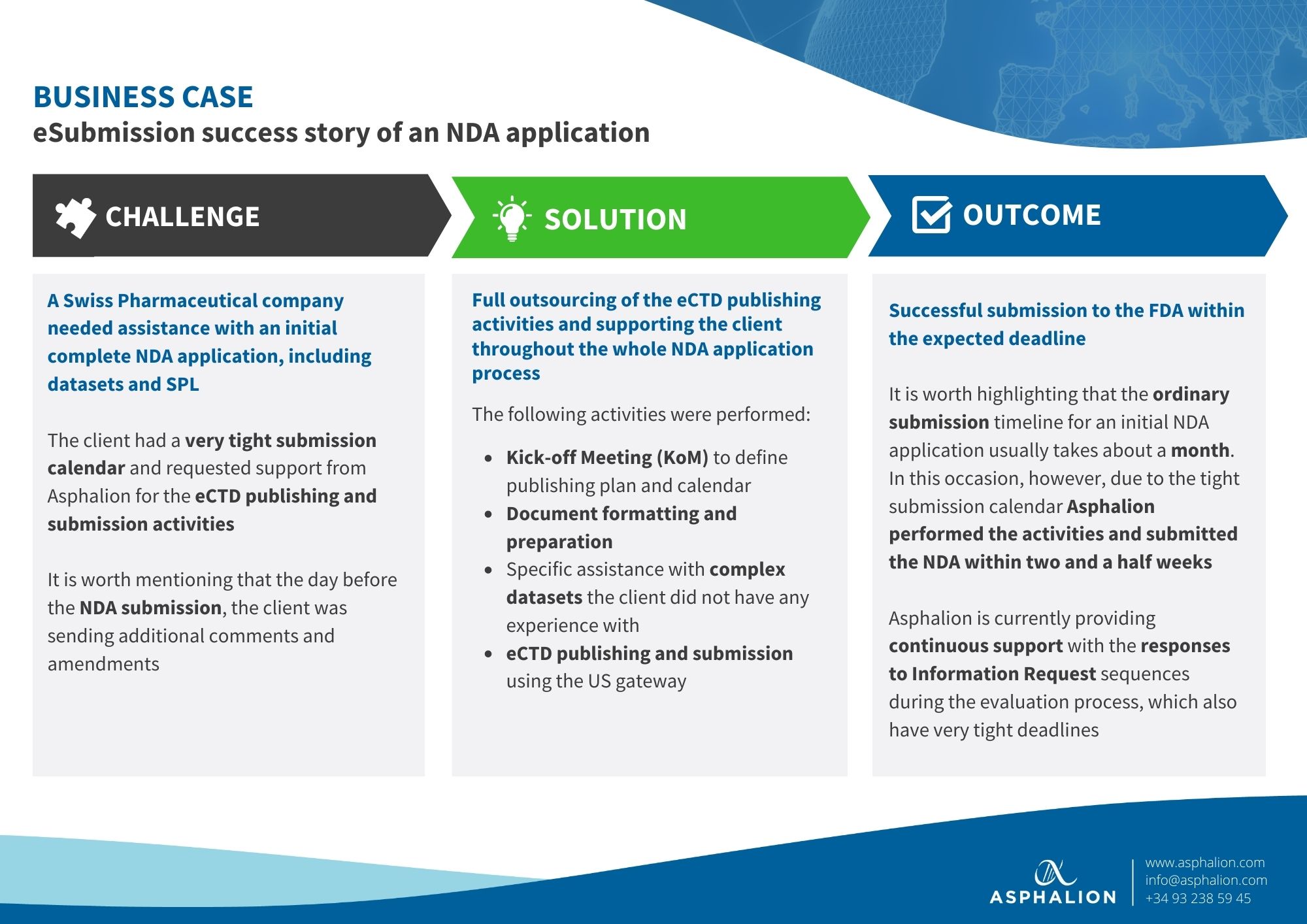
Have a look at a recent success story of how Asphalion prepared and submitted an NDA application to the FDA for a Swiss Pharmaceutical company with a very tight submission calendar.
Thanks to our extensive track record of over 20 year at Asphalion we can assist you with your eSubmissions to the FDA, EMA, etc. and with all other kind of projects.
CLICK HERE TO SEE THE CASE STUDY
For further information you can contact us at: [email protected]