Resources Hub
Welcome to the Asphalion Resources Repository
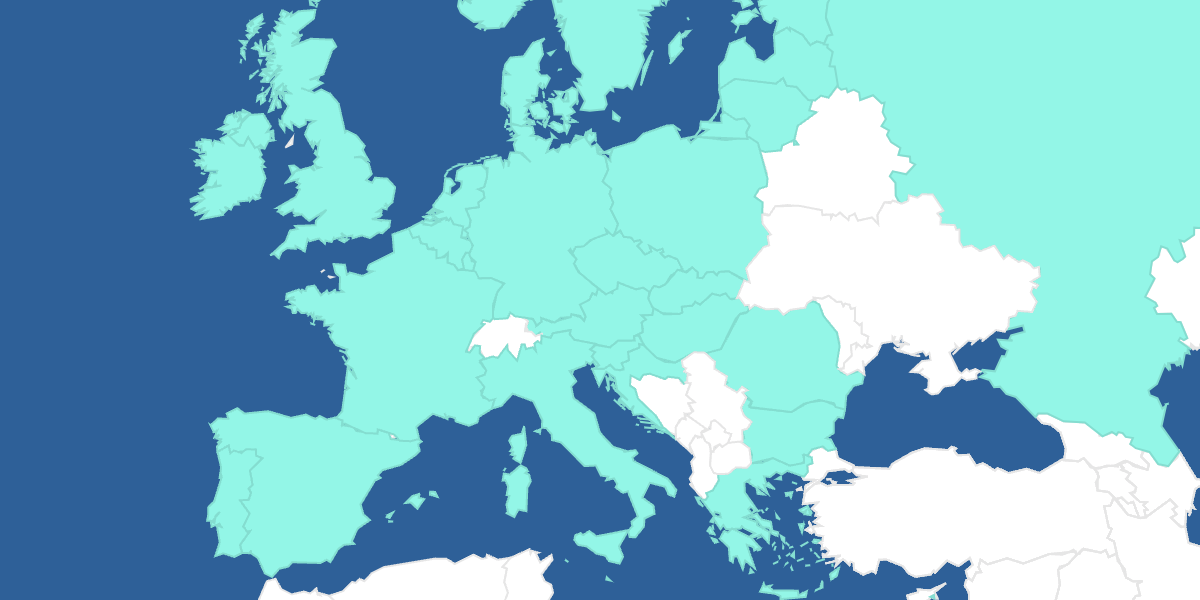
Asphanet Pharmacovigilance Interactive Map
Click on the countries to learn about the local safety requirements.
Glossary of Pharmaceutical
Regulatory Acronyms
Comprehensive glossary that provides clear definitions of pharmaceutical achronims.
Regulatory
Resources Library
Find here a collection of resources that can be useful in your day-to-day: Flyers, infographics, videos, webinars, whitepapers, etc.
We put at your disposal these useful and handy tools:
AsphaNet Video
Videos
A network of qualified partners that allows us to provide local pharmacovigilance services across Europe
EU Pharmacovigilance local contact services
Case Studies
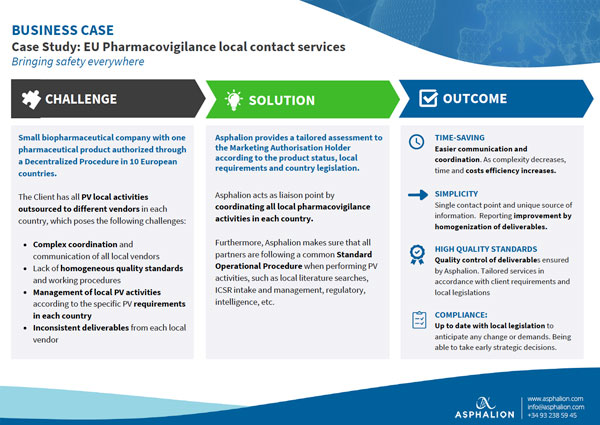
EU Pharmacovigilance local contact services case study. Small biopharmaceutical company with one
pharmaceutical product authorized through
a Decentralized Procedure in 10 European
countries.
The Client has all PV local activities
outsourced to different vendors in each
country, which poses several challenges.
Marketing Authorization Expansion in Europe
Case Studies
A case study about Marketing Authorization Expansion in Europe. A small European
Pharmaceutical company with
local activity in its country, with a
growth strategy aiming to
expand to international
markets but lacking experience
and capability in managing
complex EU procedures.