News and Events
Stay in touch with the latest news and upcoming events featuring Asphalion

PRAGMATIL | First annual progress meeting
The partners of the PragmaTIL consortium have gathered in Barcelona to hold its first annual meeting. Asphalion’s experts Montse Domingo and Nuria García Pazos attended the meeting. After this first year of work, the submission of CTA to the CTIS portal for Spain, Denmark and the Netherlands has been completed. Asphalion, as the regulatory partner, has been in charge of supervising and coordinating the regulatory path, focusing on the submission of the clinical trial and the safety management plan. During this meeting, the partners have been able to review all the work carried out to date, as well as focusing
PRAGMATIL | First annual progress meeting
The partners of the PragmaTIL consortium have gathered in Barcelona to hold its first annual meeting. Asphalion’s experts Montse Domingo and Nuria García Pazos attended the meeting. After this first year of work, the submission of CTA to the CTIS portal for Spain, Denmark and the Netherlands has been completed. Asphalion, as the regulatory partner, […]
More News & Events

New Consultancy Operations Director | André Mota
Good news at Asphalion! We are thrilled to announce that André Mota has been appointed Consultancy Operations

NEWS | High-risk medical devices pilot
EMA has launched a pilot to give scientific advice on the intended clinical development strategy and proposals for clinical investigation for certain high-risk medical devices.

WEBINAR | Clinical Trials transition to CTR/CTIS
Ready to equip yourself with first-hand information from our experts? Join us in our new free webinar,
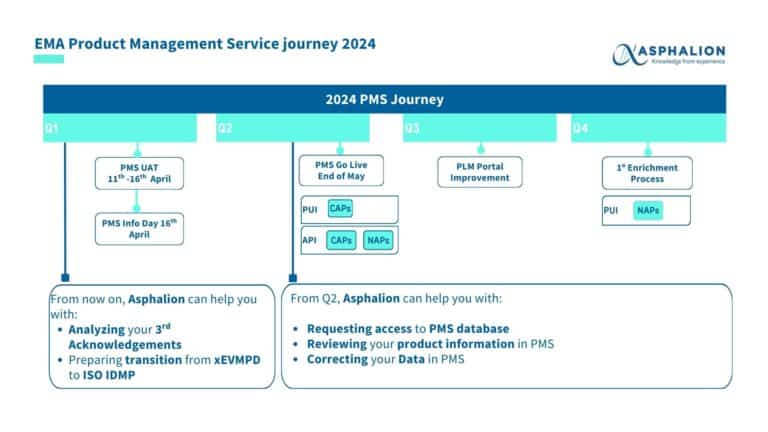
EVENT | EMA PMS Info Day
EMA is holding a new public event with the purpose to provide a comprehensive understanding of Product Management Service (PMS) and its implications on other EMA digital services with the objective to introduce the core concepts of PMS, ensuring that all attendees have the chance to understand its implementation.

EVENT | 20th Biosimilar Medicines Conference by MFE
During these two interactive days, actors of the healthcare community alongside world experts in biosimilar medicines will gather and debate the evolving biosimilar medicines landscape and emerging trends. Discussion will focus on sharing good practices and setting collective aims to achieve the depth, breadth and speed of the use of biosimilar medicines as a lever to untap their transformative value.

NEWS | New Insight on the Implementation of MDR Article 17
Article 17 of Regulation (EU) 2017/745 (Medical Device Regulation – MDR) regulates the reprocessing of single-use devices (SUDs) with relevance for the European Economic Area (EEA) which may only take place where permitted by national law and in accordance with this article.
Search or filter











