Asphalion has its own regulatory operations team. Many pharmaceutical companies across the globe rely their publishing activities on our team, which offers multidisciplinary skills and services and embrace a different bunch of services like publishing activities in different formats.
Our experienced regulatory specialists work with the latest software using self-developed project management tools to guarantee compliance with critical submission timelines.
Many of our regulatory experts have decades of experience providing support for all types of regulatory procedures and types of dossiers. Our team collaborates regularly with Health Authorities, industry experts and EMA (European Medicines Agency) SMEs (Subject Matter Experts), making Asphalion your ideal partner for the most challenging regulatory submissions.
Asphalion has its own regulatory operations team. Many pharmaceutical companies across the globe rely their publishing activities on our team, which offers multidisciplinary skills and services and embrace a different bunch of services like publishing activities in different formats.
Our experienced regulatory specialists work with the latest software using self-developed project management tools to guarantee compliance with critical submission timelines.
Many of our regulatory experts have decades of experience providing support for all types of regulatory procedures and types of dossiers. Our team collaborates regularly with Health Authorities, industry experts and EMA (European Medicines Agency) SMEs (Subject Matter Experts), making Asphalion your ideal partner for the most challenging regulatory submissions.
Regulatory Operations: eCTD Publishing and eSubmissions Solutions
Our services:
Boost your company health authorities approvals with the help of Asphalion’s Regulatory operations team. This is a comprehensive list of the publishing & submission related services that are most frequently demanded by our customers. If you have any other particular need, just let us know and we will manage it for you.
Moreover, Asphalion has a solid partnership with Extedo of more than 10 years, using their tools for publishing activities (e.g., eCTD Manager®, EURS Validator®) and providing customer support on their behalf for their regional clients.
Publish submission-ready documentation more quickly with Asphalion’s expertise. This is a comprehensive list of formatting related services that are most frequently demanded by our customers. If you have any other particular need, just let us know and we will manage it for you.
Implement regulatory affairs software right from the beginning wit our experise. This is a comprehensive list of software related services that are most frequently demanded by our customers. If you have any other particular need, just let us know and we will manage it for you.
We offer training courses on eSubmissions and Publishing with Extedo’s tools eCTDManager, EURSValidator and eSUBManager.
The content and duration are fully adaptable to the client’s needs and to the users’ knowledge level.
The training is delivered by experts in the area and includes the deliverables of the training material (i.e., manuals, guidelines, presentations, etc.) and the issuance of a training certificate.
The training methodology is mainly online, although it can also be carried out in person, as per your request. Training sessions are interactive and can be delivered both in Spanish and English, and the number of online attendees is unlimited. Additionally, a training server can be enabled for the attendees to carry out real practical cases.
This is a comprehensive list of the most demanded training topics:
Increase your productivity and reduce cost with the expertise of Asphalion’s Regulatory Operations consulting team.
Customize your Regulatory Intelligence (RI) database by subscribing to Asphalion’s Regulatory Operations newsletter, helping you to avoid resource-intensive process of gathering regulatory information:
Schedule a free meeting!
Discuss your case with our experts and receive a valuable feedback
Share
Why Asphalion?
At Asphalion, we have been at the forefront of electronic submission of regulatory information since the introduction of eCTD.
As a result of our flexible support and dedicated project management, we can produce high quality submissions within critical timelines.
We have been offering eCTD services since 2008 and, nowadays, we manage over 1,500 eCTD sequences per year.
Our ISO 9001 certification ensures traceability and secured storing.


Related Resources

Since 1st October 2022, applicants may now submit chemical structures as a single Structure-Data File format.
Are you holding a dossier in US? If the answer is YES, this brochure may be of your interest!

Asphalion ensures that eCTD submission will always follow the most updated Module 1 GCC specifications and validation rules.

IND preparation and submission business case: A small European biotech company focused on drug discovery and development interested in engaging with the FDA.
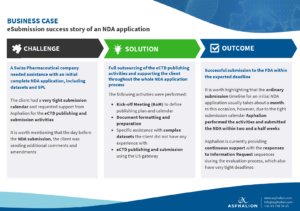
In this business case about an Asphalion eSubmission NDA, a Swiss Pharmaceutical company needed assistance with an initial complete NDA application, including datasets and SPL.

Asphalion contributions to the fight against COVID-19

EMA held the first system demo of 2024, the ninth ever held by them as part of its Agile Network Portfolio management.

Harness the power of convenience with Asphalion’s scheduling tool – a streamlined way to arrange a meeting with our Regulatory and Safety experts.

Watch Asphalion experts Carlos Domingo Palacios, Beatriz Ugalde, Adi Ickowicz, Rosa Sellabona, Álvaro Villarino, Vanesa Palau González, PhD and Daniel Langa García navigate the most critical topics influencing the Regulatory
Services
© Asphalion
Schedule here a free 30-minutes meeting with one of our consultants and tell us about your project, challenges or doubts.
We will be happy to assist you!

| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-advertisement | 1 year | Set by the GDPR Cookie Consent plugin, this cookie records the user consent for the cookies in the "Advertisement" category. |
| cookielawinfo-checkbox-analytics | 1 year | Set by the GDPR Cookie Consent plugin, this cookie records the user consent for the cookies in the "Analytics" category. |
| cookielawinfo-checkbox-necessary | 1 year | Set by the GDPR Cookie Consent plugin, this cookie records the user consent for the cookies in the "Necessary" category. |
| cookielawinfo-checkbox-others | 1 year | Set by the GDPR Cookie Consent plugin, this cookie stores user consent for cookies in the category "Others". |
| CookieLawInfoConsent | 1 year | CookieYes sets this cookie to record the default button state of the corresponding category and the status of CCPA. It works only in coordination with the primary cookie. |
| elementor | never | The website's WordPress theme uses this cookie. It allows the website owner to implement or change the website's content in real-time. |
| viewed_cookie_policy | 1 year | The GDPR Cookie Consent plugin sets the cookie to store whether or not the user has consented to use cookies. It does not store any personal data. |
| wpEmojiSettingsSupports | session | WordPress sets this cookie when a user interacts with emojis on a WordPress site. It helps determine if the user's browser can display emojis properly. |
| Cookie | Duration | Description |
|---|---|---|
| _ga | 1 year 1 month 4 days | Google Analytics sets this cookie to calculate visitor, session and campaign data and track site usage for the site's analytics report. The cookie stores information anonymously and assigns a randomly generated number to recognise unique visitors. |
| _ga_* | 1 year 1 month 4 days | Google Analytics sets this cookie to store and count page views. |
| _gat_gtag_UA_* | 1 minute | Google Analytics sets this cookie to store a unique user ID. |
| _gid | 1 day | Google Analytics sets this cookie to store information on how visitors use a website while also creating an analytics report of the website's performance. Some of the collected data includes the number of visitors, their source, and the pages they visit anonymously. |
| Cookie | Duration | Description |
|---|---|---|
| VISITOR_INFO1_LIVE | 6 months | YouTube sets this cookie to measure bandwidth, determining whether the user gets the new or old player interface. |
| VISITOR_PRIVACY_METADATA | 6 months | YouTube sets this cookie to store the user's cookie consent state for the current domain. |
| YSC | session | Youtube sets this cookie to track the views of embedded videos on Youtube pages. |
| yt-remote-connected-devices | never | YouTube sets this cookie to store the user's video preferences using embedded YouTube videos. |
| yt-remote-device-id | never | YouTube sets this cookie to store the user's video preferences using embedded YouTube videos. |
| yt.innertube::nextId | never | YouTube sets this cookie to register a unique ID to store data on what videos from YouTube the user has seen. |
| yt.innertube::requests | never | YouTube sets this cookie to register a unique ID to store data on what videos from YouTube the user has seen. |